 Breeze Medical
Breeze Medical
 Breeze Medical
Breeze Medical
 5000ml Erlenmeyer Flask Plastic PETG Flask for Shake Flask Suspension Cell Culture
5000ml Erlenmeyer Flask Plastic PETG Flask for Shake Flask Suspension Cell Culture
 PC PETG 125ml 250ml 500ml 1000ml 2800ml 5000ml Plastic Cell Culture Bottle Shake Round Bottom Erlenmeyer Flask
PC PETG 125ml 250ml 500ml 1000ml 2800ml 5000ml Plastic Cell Culture Bottle Shake Round Bottom Erlenmeyer Flask
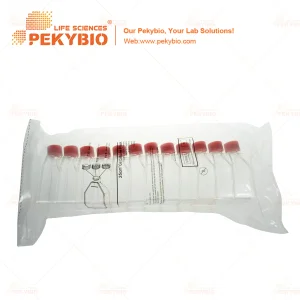 Premium Individual Cell Culture Flask for Lab Research and Test
Premium Individual Cell Culture Flask for Lab Research and Test
 Manufacturer Cell Culture Flask Sealed Cap Breathable Cap No Treated Sterile
Manufacturer Cell Culture Flask Sealed Cap Breathable Cap No Treated Sterile
As a premier Cell Culture Flasks Supplier & Exporter in Seoul, we recognize the city as more than just a capital; it is a global epicenter for biopharmaceutical innovation. From the high-tech clusters in Magok-dong to the historical research hubs in Hongneung, Seoul is driving the next generation of cell-based therapies. The demand for high-quality PETG Erlenmeyer flasks and TC-treated culture vessels has skyrocketed as South Korean firms like Samsung Biologics and Celltrion set new global standards in CDMO services.
Our role as a supplier is to bridge the gap between world-class manufacturing and the rigorous local demands of Seoul’s laboratory researchers. Whether it's for vaccine development, monoclonal antibody production, or stem cell research, our flasks provide the sterile, high-yield environment necessary for sensitive biological processes.
Supplying the Gasan Digital Complex and Seoul Bio Hub with precision instruments for regenerative medicine.
Supporting South Korea's "Bio-Health Strategy" with sterile, single-use consumables that reduce cross-contamination risk.
Strategic logistics from Seoul to North America and Europe, ensuring temperature-stable delivery of lab supplies.
The Korean biotechnology landscape is shifting towards High-Throughput Screening (HTS) and automated cell culture systems. This evolution requires flasks that are not only durable but also consistent in their surface properties. In Seoul, the move towards large-scale suspension culture has led to a surge in 5L Erlenmeyer flask usage, particularly in the production of recombinant proteins.
Furthermore, the "Smart Lab" movement in South Korean universities (like SNU and Yonsei) demands consumables that integrate seamlessly with orbital shakers and automated CO2 incubators. Our breathable membrane caps are specifically designed to maintain optimal gas exchange ratios, a critical factor for the fast-growing CHO cell lines used in Seoul’s industrial labs.
Sterility Assurance Level
Medical Grade Materials
Technical Support for Seoul Labs
Certified Quality Systems
The Magok Industrial Complex is home to numerous biotechs focusing on cell line development. Our 125ml to 1000ml Erlenmeyer flasks are the preferred choice for pilot-scale suspension cultures due to their crystal-clear PETG clarity and break-resistant properties.
Within the Seoul Bio Hub, academic researchers utilize our TC-treated T-flasks for adherent cell cultures (such as HeLa or HEK293). The specialized surface treatment ensures uniform cell attachment and optimal growth kinetics.
Seoul’s robust diagnostic sector requires massive quantities of sterile culture vessels for viral vector production. Our breathable cap designs allow for maximized oxygenation during high-speed shaking, ensuring high titers.
Hospitals like Asan Medical Center and Samsung Medical Center rely on traceable, GMP-compliant consumables for processing patient-derived cells for personalized medicine.
Ningbo Breeze Medical Co., Ltd., founded in 2011, has established itself as a leader in the medical device industry, specializing in the manufacturing of disposable medical consumables and laboratory instruments. Committed to providing the highest quality products, the company has invested in state-of-the-art infrastructure, including a cleanroom, an automatic injection molding workshop, and a support production line that meets the strictest GMP (Good Manufacturing Practice) standards.
Since its inception, Ningbo Breeze Medical has adhered to a business philosophy centered around quality and innovation. The company follows a stringent quality management and control system that complies with international standards, ensuring that its products are reliable and safe for use in various medical applications in the Seoul region and beyond. We have earned prestigious certifications such as ISO 9001:2008, ISO 13485:2003, and CE.
Continuous investment in developing new technologies for the Korean medical and laboratory sectors.
Our team of highly skilled engineers provides 24/7 support for all Seoul-based projects.
Compliance with CE, ISO 13485, and ISO 9001 guarantees reliable results in every experiment.
 225 Cm² Breathable Cap No Treated Sterile N Design Cell Culture Flask
225 Cm² Breathable Cap No Treated Sterile N Design Cell Culture Flask
 Biobase China Factory Direct Supply Breathable Membrane Cap 500ml Triangular Cell Culture Shake Flask
Biobase China Factory Direct Supply Breathable Membrane Cap 500ml Triangular Cell Culture Shake Flask
 High Quality Tissue Cell Culture Flask Treated Cell Culture Flask with Standard
High Quality Tissue Cell Culture Flask Treated Cell Culture Flask with Standard
 175 Cm² Cell Culture Flask Seal Cap Tc Treated Sterile N Design
175 Cm² Cell Culture Flask Seal Cap Tc Treated Sterile N Design
 PETG Pet Plastic 125ml-1000ml Clear Tissue Cell Culture Lab Conical Erlenmeyer Flask
PETG Pet Plastic 125ml-1000ml Clear Tissue Cell Culture Lab Conical Erlenmeyer Flask
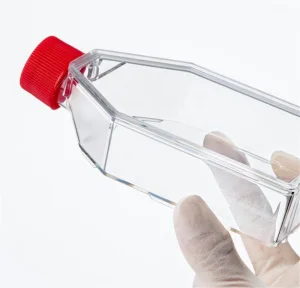 Culture Flask Plastic Tissue Cell Culture Flask Radical Manufacturer
Culture Flask Plastic Tissue Cell Culture Flask Radical Manufacturer
 Medical Science Cell Tissue Culture Bottle Flask 250ml Plastic Sterile Flask
Medical Science Cell Tissue Culture Bottle Flask 250ml Plastic Sterile Flask
 Durable PETG Shake Flask for Laboratory Cell Culture Applications
Durable PETG Shake Flask for Laboratory Cell Culture Applications