 Breeze Medical
Breeze Medical
 Breeze Medical
Breeze Medical
The global market for Viral Transport Media (VTM) has transitioned from an emergency pandemic response to a foundational component of modern molecular diagnostics. As a primary Viral Transport Medium Manufacturer, we observe a steady surge in demand for high-quality transport solutions that ensure nucleic acid stability during transit.
Current industry trends focus on room-temperature stability and the shift toward inactivated media, which reduces biohazard risks during handling. Custom OEM VTM solutions are increasingly integrated with automated extraction platforms to streamline laboratory workflows.
International procurement agents and government health agencies are moving away from generic suppliers toward ISO 13485 certified manufacturers. Reliability, supply chain resilience, and the ability to scale production rapidly are now the top criteria for global VTM sourcing.
Ningbo Breeze Medical Co., Ltd., founded in 2011, has established itself as a leader in the medical device industry, specializing in the manufacturing of disposable medical consumables and laboratory instruments. Committed to providing the highest quality products, the company has invested in state-of-the-art infrastructure, including a cleanroom, an automatic injection molding workshop, and a support production line that meets the strictest GMP (Good Manufacturing Practice) standards. In addition, the company has set up a Product Quality Testing Center to ensure the reliability and performance of all its products.

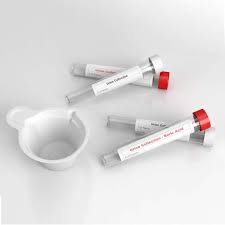
Our VTM formulations are engineered for the preservation of clinical specimens containing viruses, including Influenza, SARS-CoV-2, and other respiratory pathogens. As a Custom VTM Supplier, we offer two primary types of media:
By leveraging our OEM capabilities, clients can customize tube sizes (5ml, 10ml), media volume (1.5ml, 3ml), and even the chemical composition to meet specific diagnostic kit requirements. Our expertise in injection molding ensures that the transport tubes are leak-proof and compatible with standard centrifuges.
Our portfolio extends beyond VTM to provide a comprehensive ecosystem for healthcare providers:
In the global medical supply chain, China stands as the powerhouse for medical consumables. Choosing Ningbo Breeze Medical as your partner offers distinct advantages:
Scientific research is a key pillar of Breeze Medical’s innovation. The company continuously invests in developing new technologies and products that contribute to improved healthcare.
It boasts a team of highly skilled technical support engineers who provide 24/7 assistance to address any customer inquiries, ensuring professional and timely service.
Ningbo Breeze Medical Co., Ltd. has significantly expanded its presence in both domestic and international markets, reflecting growing trust in the brand.