 Breeze Medical
Breeze Medical
 Breeze Medical
Breeze Medical
 Sterile Microbiology Bacteria Cell Inoculating Loop 1UL
Sterile Microbiology Bacteria Cell Inoculating Loop 1UL
 Bioland High Quality Microcentrifuge Tube Low Retention Sterile 1.5ml
Bioland High Quality Microcentrifuge Tube Low Retention Sterile 1.5ml
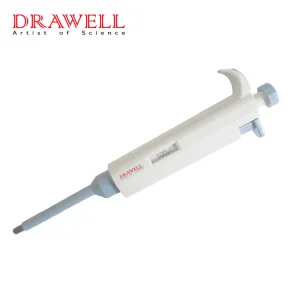 Laboratory Plastic Pipette Automatic Measuring Pipette Micro Pipette
Laboratory Plastic Pipette Automatic Measuring Pipette Micro Pipette
 Lab Use 50ml 250ml Tissue Culture Bottle Graduated Plastic Cell Culture Flask
Lab Use 50ml 250ml Tissue Culture Bottle Graduated Plastic Cell Culture Flask
 10ml Sterile Serological Pipette Dnase/Rnase Free
10ml Sterile Serological Pipette Dnase/Rnase Free
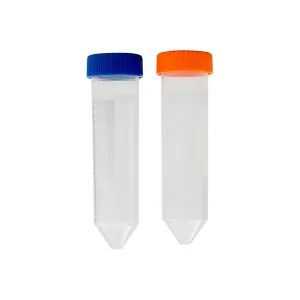 Lab Consumables Conical Bottom 50ml Falcon Centrifuge Tubes
Lab Consumables Conical Bottom 50ml Falcon Centrifuge Tubes
 1000ml PETG Laboratory Flat Bottom Erlenmeyer Flask
1000ml PETG Laboratory Flat Bottom Erlenmeyer Flask
 Wholesale Laboratory Test Tube Microcentrifuge Tube
Wholesale Laboratory Test Tube Microcentrifuge Tube
In the modern high-throughput laboratory environment, the humble pipette holder has transitioned from a simple storage accessory to a critical component of ergonomic health, contamination control, and workflow efficiency. As a leading OEM/ODM Pipette Holders Manufacturer, we recognize that global procurement demands are shifting. It is no longer sufficient to provide a static stand; today's laboratories require modular, antimicrobial, and chemically resistant systems that integrate seamlessly with advanced liquid handling protocols.
This white paper explores the macro-industrial landscape of pipette management, detailing how technical innovations in polymer science and precision injection molding are setting new standards for the Life Sciences industry. From ISO-certified cleanroom production to localized global support, the roadmap for pipette holder development is increasingly driven by the need for customized, brand-agnostic solutions that serve both clinical and research sectors.
Addressing the global demand for "Just-in-Time" delivery of lab consumables. Our logistics network ensures that large-scale ODM orders reach international hubs with full regulatory documentation.
Implementing silver-ion infused plastics and UV-resistant coatings to ensure that pipette holders act as a barrier against cross-contamination in sterile environments.
Custom design solutions that reduce repetitive strain injury (RSI) by optimizing the angle of access and stability of multi-channel pipette storage.
The rise of smart labs. We are developing holders with RFID tracking capabilities to monitor instrument calibration cycles and usage data.
Transitioning toward recyclable medical-grade polymers and reducing carbon footprints through high-efficiency, automated injection molding processes. Our "Circular Lab" initiative focuses on life-cycle management of plastic consumables.
Phase 1: Advanced Material Science
Utilization of high-density polyethylene (HDPE) and reinforced polypropylene (PP) for superior chemical resistance against common lab solvents and disinfectants.
Phase 2: Precision Engineering
Implementing 0.01mm tolerance in injection molding to ensure universal fitment for all major pipette brands (Gilson, Eppendorf, Rainin, etc.).
Phase 3: Digital & Modular Design
Developing 3D-printable prototypes for rapid OEM iteration, allowing clients to test ergonomic fit in 48 hours.
Driving innovation through GMP-compliant manufacturing and ISO 13485 standards.
Ningbo Breeze Medical Co., Ltd., founded in 2011, has established itself as a leader in the medical device industry, specializing in the manufacturing of disposable medical consumables and laboratory instruments. Committed to providing the highest quality products, the company has invested in state-of-the-art infrastructure, including a cleanroom, an automatic injection molding workshop, and a support production line that meets the strictest GMP (Good Manufacturing Practice) standards. In addition, the company has set up a Product Quality Testing Center to ensure the reliability and performance of all its products.

Since its inception, Ningbo Breeze Medical has adhered to a business philosophy centered around quality and innovation. The company follows a stringent quality management and control system that complies with international standards, ensuring that its products are reliable and safe for use in various medical applications. As a result, Breeze Medical has earned prestigious certifications such as ISO 9001:2008, ISO 13485:2003, and CE, assuring that its products meet global quality and safety standards.
Breeze Medical specializes in the manufacturing of a wide range of medical products, including disposable medical consumables and laboratory instruments used in hospitals, clinics, and research centers. Key capabilities include:
The company’s modern production infrastructure optimizes efficiency and precision. Its success in global markets demonstrates its ability to offer innovative, high-quality products to customers worldwide. Furthermore, a team of highly skilled technical support engineers provides 24/7 assistance to address any customer inquiries, ensuring professional service at all times.
Navigating the complex regulatory landscape of the medical and laboratory sectors is a core competency of our OEM/ODM service. We provide comprehensive documentation including CE Certification ISO 13485 Free Sales Certificates and RoHS Compliance.
Our localized support model includes technical warehousing in key regions and a dedicated regulatory affairs team that assists partners in registering products with local health authorities (e.g., FDA, NMPA, EMA). This ensures that our pipette holder supply chain is not only efficient but also fully compliant with the evolving standards of the global healthcare industry.
We primarily use medical-grade ABS and Polypropylene (PP). For specialized ODM projects, we can incorporate antimicrobial additives or heat-resistant polymers for autoclave compatibility.
Yes. We offer various customization options including laser-engraved logos, custom color matching (Pantone), and bespoke packaging designs to align with your brand identity.
Prototyping takes 7-10 days, while the production of a new injection mold typically requires 30-45 days, followed by rigorous quality testing before mass production begins.
While we specialize in high-volume manufacturing, we offer flexible MOQ (Minimum Order Quantity) tiers to support long-term strategic partnerships and innovative laboratory startups.
Our engineers utilize high-precision 3D scanning and CAD modeling to create universal or brand-specific inserts, ensuring a secure and stable fit for all major pipette models.
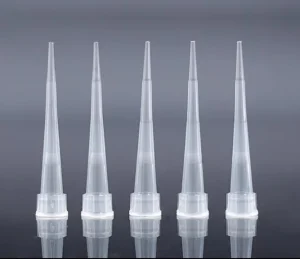 Universal Sterile Plastic Disposable Micro Pipette Tips with Filter
Universal Sterile Plastic Disposable Micro Pipette Tips with Filter
 High Quality Tissue Cell Culture Flask Treated
High Quality Tissue Cell Culture Flask Treated
 E-Beam Sterile Cell Culture Flask for Laboratories
E-Beam Sterile Cell Culture Flask for Laboratories
 Disposable Hospital Graduated Measuring Pipette 10ml
Disposable Hospital Graduated Measuring Pipette 10ml
 Eight-Channel and Twelve-Channel Volumetric Adjustable Pipette
Eight-Channel and Twelve-Channel Volumetric Adjustable Pipette
 Medical Use 3-Layers Plastic Pathology Ziplock Specimen Bag
Medical Use 3-Layers Plastic Pathology Ziplock Specimen Bag
 Individually Wrapped 1ml 2ml 5ml 10ml 25ml Serological Pipette
Individually Wrapped 1ml 2ml 5ml 10ml 25ml Serological Pipette
 Adjustable Electric Pipette Autoclavable Transfer Pipettor
Adjustable Electric Pipette Autoclavable Transfer Pipettor