 Breeze Medical
Breeze Medical
 Breeze Medical
Breeze Medical
 Sterile Microbiology Bacteria Cell Inoculating Loop 1UL
Sterile Microbiology Bacteria Cell Inoculating Loop 1UL
 High Quality Laboratory Supplies Plastic Disposable Sterile Inoculating Loops
High Quality Laboratory Supplies Plastic Disposable Sterile Inoculating Loops
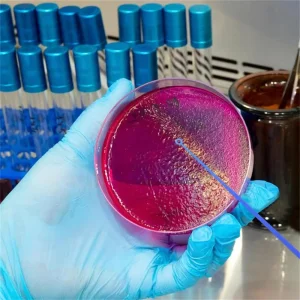 Laboratory Disposable Plastic Sterile Inoculating Loop Inoculum 1UL 10UL
Laboratory Disposable Plastic Sterile Inoculating Loop Inoculum 1UL 10UL
 Plastic Disposable Inoculating Loops 10UL Inoculation Loop Sterilizer
Plastic Disposable Inoculating Loops 10UL Inoculation Loop Sterilizer
 Disposable Sterile Culture Inoculating Loop and Needle Inoculum 1UL 10UL
Disposable Sterile Culture Inoculating Loop and Needle Inoculum 1UL 10UL
 Disposable Laboratory Rigid and Flexible Inoculating Loop 1UL, 10UL, Needle
Disposable Laboratory Rigid and Flexible Inoculating Loop 1UL, 10UL, Needle
 Laboratory Plastic Disposable Bacterial 1UL 10UL Inoculation Loop Sterilizer
Laboratory Plastic Disposable Bacterial 1UL 10UL Inoculation Loop Sterilizer
 Inoculating Loops Wholesale Clinical Research Laboratory 1UL 10UL
Inoculating Loops Wholesale Clinical Research Laboratory 1UL 10UL
In the contemporary landscape of clinical diagnostics and life science research, Inoculating Needles and Loops serve as the fundamental tools for microbial cultivation. As global health awareness rises and the pharmaceutical industry expands, the demand for high-precision, contaminant-free inoculation tools has reached an all-time high.
Currently, the market is shifting from traditional reusable metal loops to disposable plastic medical-grade polystyrene alternatives. This transition is driven by the critical need to eliminate cross-contamination and the time-consuming sterilization process involving Bunsen burners. From the massive diagnostic hubs in North America and Europe to the burgeoning biotech sectors in Southeast Asia, the reliability of a manufacturer determines the accuracy of laboratory results worldwide.
Modern labs are moving towards high-throughput robotic systems. Manufacturers are now designing needles with precise rigidity and uniform dimensions to fit seamlessly into automated plate streaking machines.
The use of ultra-smooth, hydrophobic medical-grade polymers ensures that the exact volume (1uL or 10uL) is transferred without surface tension interference, a key factor in quantitative microbiology.
Industry-standard color coding (e.g., Blue for 10uL, Clear/White for 1uL) is becoming universal to minimize human error in fast-paced clinical environments.
When B2B buyers—ranging from large-scale hospital chains to academic research distributors—search for the Top 10 Inoculating Needles Manufacturer, they look beyond just price. The primary concern is Reliability and Traceability.
Key Sourcing Criteria:
Ningbo Breeze Medical Co., Ltd., founded in 2011, represents the pinnacle of Chinese medical manufacturing. We combine cost-efficiency with uncompromising quality standards that rival any Western competitor.
Our facility features a state-of-the-art cleanroom and an automatic injection molding workshop. Every inoculating needle is produced in a controlled environment to ensure zero biological load before sterilization.
From mold design to final packaging, we control the entire supply chain. Our Product Quality Testing Center monitors every batch for flexibility, smoothness, and volume accuracy.
With ISO 9001:2008, ISO 13485:2003, and CE certifications, our products are pre-approved for the most demanding markets in Europe and North America.
Ningbo Breeze Medical Co., Ltd. focuses on providing the highest quality products. Our automated production line for vacuum blood collection tubes and laboratory instruments has positioned us as a key supplier in the healthcare sector. We don't just sell products; we provide 24/7 technical support through our team of highly skilled engineers.
Inoculating needles are used differently across various regions and sectors:
 High Quality Disposable Plastic Sterile 1UL 10UL Inoculation Loop
High Quality Disposable Plastic Sterile 1UL 10UL Inoculation Loop
 Lab Disposable Plastic Sterile 1UL Inoculating Loops 10UL
Lab Disposable Plastic Sterile 1UL Inoculating Loops 10UL
 Inoculating Loops 1UL 10UL Lab Smear Loop Inoculation Rod
Inoculating Loops 1UL 10UL Lab Smear Loop Inoculation Rod
 Manufacturer Mini Plastic Inoculating Loop with Individual Packaging
Manufacturer Mini Plastic Inoculating Loop with Individual Packaging
 Medical Grade Polystyrene Wholesale 1UL Inoculating Loops Needles
Medical Grade Polystyrene Wholesale 1UL Inoculating Loops Needles
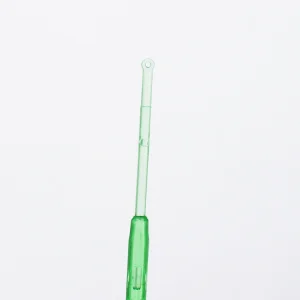 1UL/10UL Plastic Inoculation Loop Sterilizer with Needle Flexible
1UL/10UL Plastic Inoculation Loop Sterilizer with Needle Flexible
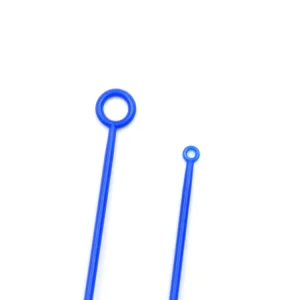 Professional Safety Heated 10UL Sterile Disposable Inoculation Loops
Professional Safety Heated 10UL Sterile Disposable Inoculation Loops
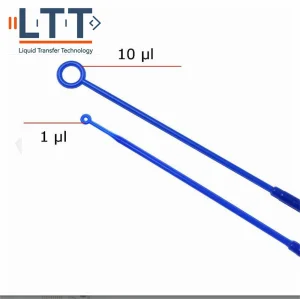 Disposable Plastic 1UL 10UL Inoculation Loop
Disposable Plastic 1UL 10UL Inoculation Loop