Breeze Medical
Breeze Medical
 Breeze Medical
Breeze Medical
Ningbo Breeze Medical Co., Ltd., founded in 2011, has established itself as a leader in the medical device industry, specializing in the manufacturing of disposable medical consumables and laboratory instruments. Committed to providing the highest quality products, the company has invested in state-of-the-art infrastructure, including a cleanroom, an automatic injection molding workshop, and a support production line that meets the strictest GMP (Good Manufacturing Practice) standards. In addition, the company has set up a Product Quality Testing Center to ensure the reliability and performance of all its products.

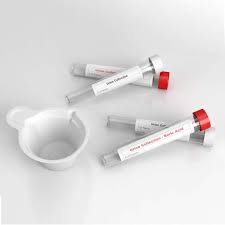
The global demand for cryovials has witnessed exponential growth, driven by the rapid expansion of biotechnology, personalized medicine, and global biobanking initiatives. As the world pivots towards advanced cell therapies and vaccine research, the necessity for ultra-low temperature storage solutions has never been more critical. Cryovials are no longer just simple plastic tubes; they are sophisticated vessels designed to protect the integrity of biological samples at temperatures as low as -196°C.
In the current industrial landscape, China has emerged as a powerhouse for laboratory consumables. Factories like Ningbo Breeze Medical are at the forefront, blending cost-efficiency with high-end manufacturing technologies that rival Western counterparts. The global supply chain now relies heavily on Chinese exporters to provide the volume and quality required for large-scale clinical trials and diagnostic networks.
With the rise of genomic research, biobanks require millions of cryovials annually. Our products ensure long-term sample viability with superior sealing technology.
The delicate nature of stem cells and CAR-T cells demands containers that are free from DNase, RNase, and pyrogens, a standard we strictly uphold.
Modern labs use robotic systems. Our cryovials are designed with standardized dimensions to fit perfectly into automated liquid handling and storage systems.
International buyers from North America, Europe, and Southeast Asia are increasingly looking for reliable OEM/ODM partners in China. Key procurement criteria include:
Used in hospitals for storing patient serum, plasma, and whole blood samples for longitudinal studies.
Essential for drug discovery phases where thousands of chemical compounds and biological reagents must be archived.
Ensuring the chain of custody and biological integrity of DNA evidence collected from crime scenes.
Preservation of plant germplasm and animal genetic material to ensure global food security and biodiversity.
As a premier China Cryovials Supplier, our advantages lie in our integrated approach to manufacturing and quality control. We don't just produce plastic; we engineer medical solutions.
Our automatic injection molding workshop ensures high precision and consistency across every batch.
Production takes place in controlled environments to eliminate any risk of biological contamination.
Holding ISO 9001, ISO 13485, and CE certifications, we meet the regulatory demands of any market.
Scientific research is a key pillar of Breeze Medical’s innovation. The company continuously invests in developing new technologies and products that contribute to improved healthcare. Additionally, it boasts a team of highly skilled technical support engineers who provide 24/7 assistance to address any customer inquiries or needs, ensuring professional and timely service at all times.
Our processing capacity allows us to manufacture products in large volumes without compromising on quality. Its modern production infrastructure includes advanced equipment that optimizes efficiency and precision in the manufacturing process. Additionally, the company has implemented automated production lines that ensure consistent quality and the ability to meet global market demands.