 Breeze Medical
Breeze Medical
 Breeze Medical
Breeze Medical
The topic of VTM kits, or Viral Transport Medium kits, is crucial in the field of virology and diagnostics. As healthcare professionals increasingly rely on these kits for the transport of clinical specimens, a pressing question arises: What is the shelf life of VTM kits? According to studies from the CDC and WHO, maintaining the integrity of samples is vital for accurate testing and diagnosis.
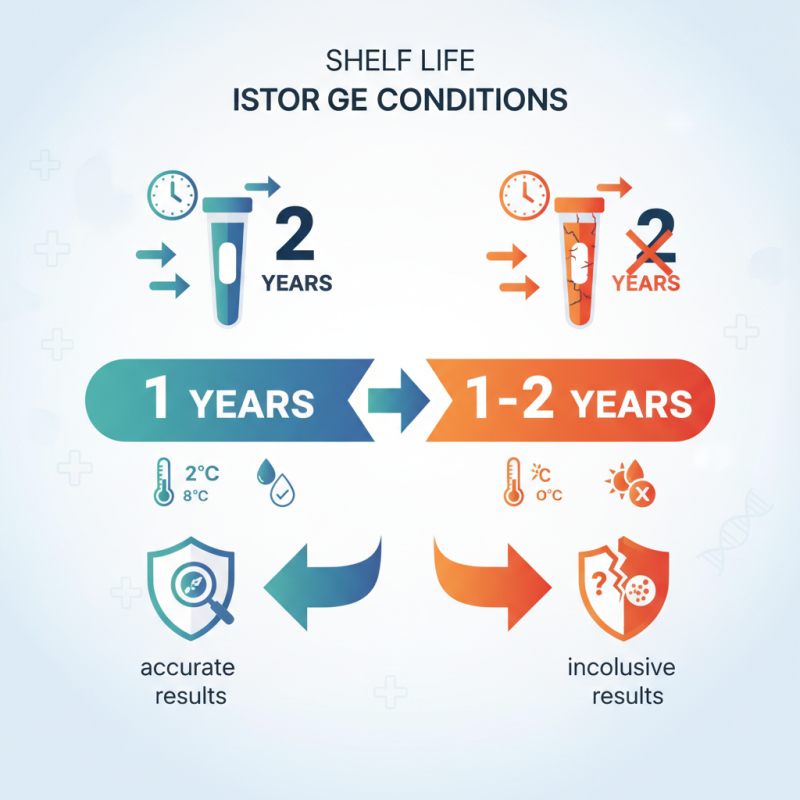
Typically, VTM kits have a shelf life of about one to two years, but this can vary based on storage conditions. Proper storage is essential. Exposure to extreme temperatures or humidity can reduce their efficacy significantly. Unfortunately, many facilities may overlook these details, risking the quality of their tests.
Monitoring expiration dates is not just a formality; it’s a matter of reliable diagnosis. An expired VTM kit might lead to inconclusive results and misdiagnoses. Awareness and proper management of these kits are necessary to uphold the standards of patient care and public health responses. Understanding the shelf life of VTM kits allows for better planning and resource allocation in healthcare settings.
Viral transport medium (VTM) kits play a crucial role in transporting clinical samples. These kits ensure that viruses remain viable and can be accurately tested. According to the Centers for Disease Control and Prevention (CDC), proper use of VTM kits is vital for effective infectious disease diagnosis. The stability of VTM can be impacted by various factors, including temperature and storage conditions. Research shows that most VTM kits have a shelf life ranging from 6 to 12 months.
Different types of VTM kits have unique storage requirements. Some need to be refrigerated, while others can be stored at room temperature. Understanding these conditions is essential for maintaining sample integrity. It is concerning that many professionals may overlook these guidelines. If VTM kits are stored improperly, test results can be compromised, leading to misdiagnosis or ineffective treatment.
**Tips:** Always check the expiration date. Ensure that VTM kits are kept in a stable environment. Document the storage conditions regularly. Make sure to discard expired VTM kits appropriately. Taking these steps boosts reliability in testing and diagnostics.
This bar chart illustrates the percentage of VTM kits that expire within different time intervals. It highlights the importance of monitoring VTM kit expiration to ensure effective sample collection and testing.
When it comes to the shelf life of Viral Transport Medium (VTM) kits, several factors play a crucial role. The primary component, the medium itself, needs to be stored correctly. Ideal temperature ranges are essential. Exposure to high temperatures can lead to degradation. Additionally, light exposure may negatively impact the viability of the contained substances.
Another important consideration is how the kits are sealed. Proper sealing prevents contamination and maintains sterility. Even minor breaches can alter the effectiveness of the medium. The expiration date on the packaging provides a guideline. However, real-world conditions can affect longevity. Regular checks are needed to ensure the kits remain usable. Sometimes, the kits might appear fine but may no longer work as intended.
Environmental factors also contribute significantly to shelf life. Humidity levels may cause kits to degrade quicker. It’s crucial to monitor storage conditions. An overlooked detail can lead to wasted resources and compromised samples. Understanding these factors can help ensure effective use of VTM kits. Maintaining awareness and adhering to guidelines will ultimately support better outcomes in testing.
The expiration dates for Viral Transport Medium (VTM) kits can vary significantly. Typically, these kits have a shelf life of 12 to 24 months. However, this can depend on the manufacturer and storage conditions. It’s crucial to check the expiration date printed on each kit. Storing VTM kits in a cool, dry place can extend their usability.
Many healthcare professionals may overlook expiration dates. They might believe that as long as the kits look fine, they are usable. This belief can lead to potential issues. Using expired kits can compromise sample integrity, affecting test results. It is essential to stay aware of the expiration timelines. Regularly checking inventory and disposing of outdated kits can prevent mishaps.
In summary, understanding when VTM kits expire is vital for accurate testing. Awareness of their shelf life can enhance laboratory efficiency. Regular training for staff on the importance of checking expiration dates is beneficial. Ultimately, vigilance in monitoring these details ensures better patient care and accurate diagnostics.
Choosing the right storage conditions is vital for maximizing the longevity of VTM kits. These kits typically contain viral transport medium, which can degrade if not stored properly. Ideally, VTM kits should be kept in a cool, dark place. Temperatures between 2°C and 8°C are often recommended. Exposing them to extreme heat or direct sunlight can cause significant deterioration.
Humidity also plays a crucial role. High humidity can compromise the integrity of the kit. Inadequate sealing or moisture exposure can lead to contamination. It is advisable to check the packaging for any visible damages or leaks before use.
Regularly monitoring storage conditions can prove challenging but necessary. Some users may overlook expiration dates, leading to the potential for compromised samples. It is important for laboratories to maintain inventory logs. This practice encourages responsible usage and timely disposal of expired products. Understanding these nuances can contribute to more reliable testing outcomes.
When dealing with expired VTM kits, understanding their reliability is vital. Even if they are past their expiration date, some kits may still yield useful results. However, the degradation of materials can affect sample preservation. Protein breakdown and loss of sterility are concerns that you should keep in mind. It is essential to assess each kit individually based on storage conditions and time since the expiration.
Best practices for using expired VTM kits start with careful inspection. Check for any visible signs of damage or leakage. If the packaging appears compromised, it’s wise to discard the kit. The sterility of the sample is paramount. Additionally, you should consider the kind of tests being performed. If results are crucial, using fresh kits is recommended. However, in a pinch, expired kits may suffice. Document every step when utilizing expired kits, noting dates and any anomalies observed. This diligence will help maintain a record of reliability, even under less-than-ideal circumstances.
| Item | Shelf Life (Months) | Common Expiration Causes | Best Practices for Use |
|---|---|---|---|
| VTM Kit A | 12 | Improper storage, temperature fluctuations | Store in a cool, dry place; check for leaks |
| VTM Kit B | 18 | Constituent degradation, light exposure | Avoid direct sunlight; monitor expiry dates |
| VTM Kit C | 24 | Moisture contamination, usage beyond limits | Seal tightly after use; review compatibility |
| VTM Kit D | 8 | Chemical instability, improper handling | Follow handling instructions; discard if expired |
: VTM kits transport clinical samples and keep viruses viable for accurate testing.
Most VTM kits have a shelf life of 6 to 12 months.
VTM kits should be stored between 2°C and 8°C, away from sunlight and heat.
Improper storage can compromise test results, leading to misdiagnosis or ineffective treatment.
High humidity can damage the kit and increase the risk of contamination.
Regularly check expiration dates and monitor storage conditions. Dispose of expired kits properly.
Inspect the packaging for damages or leaks before use to ensure integrity.
Documenting helps maintain sample integrity and encourages timely disposal of expired products.
Some VTM kits can be stored at room temperature, but others need refrigeration.
Overlooking guidelines may lead to unreliable testing outcomes and impact patient care.
Viral Transport Medium (VTM) kits play a crucial role in the collection and transport of viral specimens for testing. The shelf life of VTM kits, a key aspect to consider, is influenced by several factors including the type of reagents used, packaging, and storage conditions. Generally, the typical expiration dates for VTM kits range from six months to two years, depending on these variables.
To maximize the longevity of VTM kits, optimal storage conditions such as maintaining a consistent temperature and avoiding direct sunlight are essential. Additionally, understanding best practices for the usage of expired kits is important, as they may still retain some efficacy, although their reliability cannot be guaranteed. Overall, knowing what is the shelf life of VTM kits and adhering to recommended storage practices can ensure the effectiveness of these vital tools in viral testing.
